 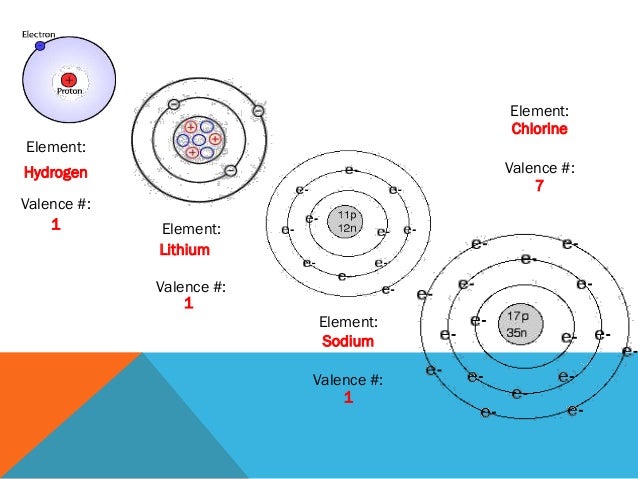
In the second period elements, the two electrons in the \(1s\) sublevel are called inner-shell electrons and are not involved directly in the element's reactivity or in the formation of compounds. How are valence electrons used to determine an atoms reactivity?! Describe the octet rule. 
Atoms for elements in the main element groups prefer to have up to eight valence electrons. How do valence electrons determine reactivity of an atom. In a reaction, one atom may accept electrons from another atom. The octet rule is a chemical rule of thumb that reflects the theory that main group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas.The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens, but also to metals such as sodium or magnesium. These elements or materials are called semiconductor. Atoms usually engage in chemical reactions which will end in complete outer electron shells. Answers (1) Jimmy Collier 27 October, 23:08. The Group 3 atoms have 3 valence electrons. The Halogens, having 7 valence electrons, achieve a full outer level by gaining one electron. In addition to that, the amount of valence electrons in the outer shell determines how atoms interact with one another. Lone pairs, unpaired electrons, and single, double, or triple bonds are used to indicate where the valence electrons are located around the atom. An atom has to gain, lose or share a particular number of electrons from its outermost orbit to obtain complete octet.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
